This recorded webinar covers the introduction of the adalimumab biosimilar Amgevita in New Zealand, and what it means for primary care. Specialist Dr David Liew shares his experiences of changing patients from biologics to biosimilars in Australia, and Andrea Copeland provides some local context.
Transition to adalimumab biosimilar

Amgevita is fully funded in New Zealand from 1 March 2022 subject to Special Authority criteria and Humira will remain available only under specific circumstances.
Key points
- The adalimumab reference product, Humira, will soon be replaced with the biosimilar, Amgevita.
- Patient access to adalimumab will be broader with the biosimilar, with no dosage restrictions and a two-year Special Authority renewal period.
- Conversations about changing from Humira to Amgevita should be initiated early and framed positively; stable patients may be transitioned in primary care.
- To avoid inadvertent substitution, always use brand names when discussing, prescribing and dispensing biological medicines.
Introduction
Biological medicines have markedly changed the prognoses for many chronic and disabling inflammatory and immunological conditions and cancers. Their use in New Zealand is increasing and, as patents expire on reference or originator “biologics”, the opportunity arises for competitor manufacturers to list biosimilars – highly similar versions of approved biological medicines.1,2
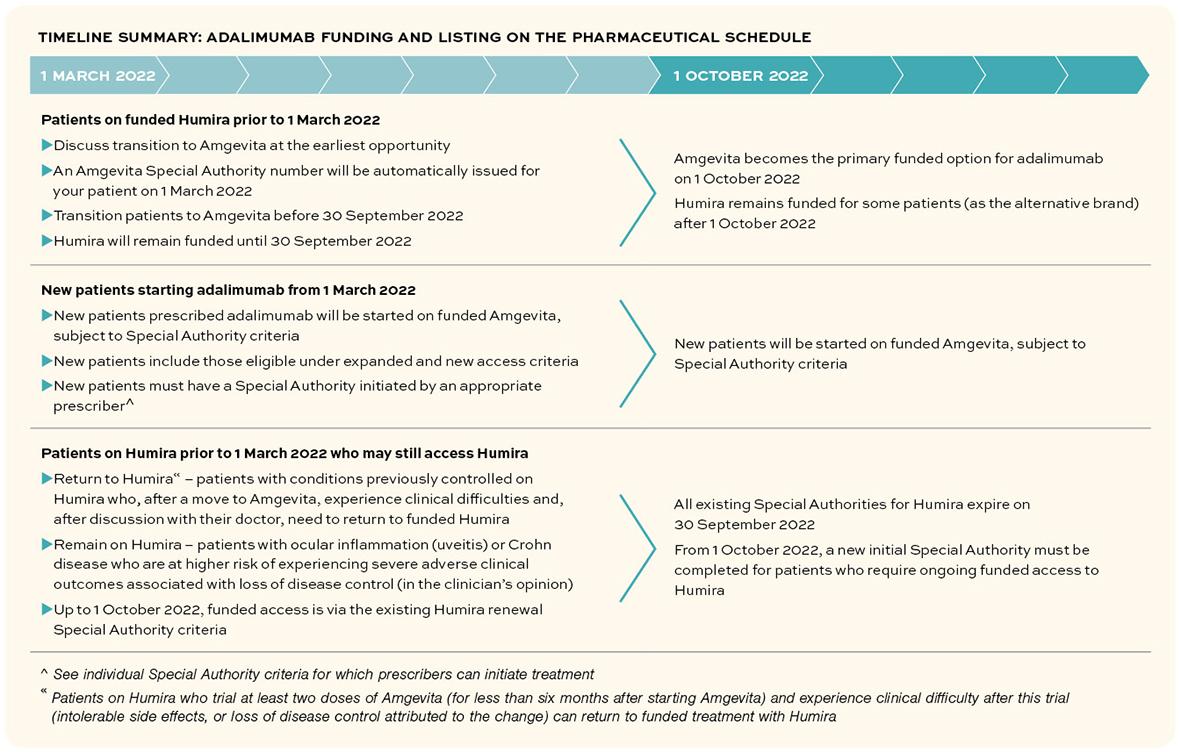
In November 2021, Pharmac announced changes to widen access to the anti-tumour necrosis factor alpha (TNF-α inhibitor) adalimumab. A biosimilar adalimumab (Amgevita) will be funded from 1 March 2022, with the reference product (Humira) remaining available only under specific circumstances.3
Many patients already stable on Humira have repeat prescriptions and Special Authority renewals performed in primary care. These patients can be transitioned to Amgevita in primary care during a seven-month window in which both brands are funded.3
From 1 October 2022, Amgevita becomes the primary funded option for adalimumab.3 During the transition window and beyond, exceptions are in place for continued funded use of Humira for certain patients who need to remain on, or move back to, the reference medicine.3
For busy primary healthcare professionals aiming to keep abreast of developments in biological medicines and the advances in treatment they provide, the large volume of new information and the addition of biosimilars into the prescribing mix can be daunting.
This article summarises the 2022 changes regarding adalimumab funding and access in New Zealand. It also provides some background and resources to foster confidence in biosimilars, and references real-world data from studies of patients changing from Humira to Amgevita.
Adalimumab - Humira and Amgevita
Adalimumab is a human TNF-α monoclonal antibody that acts as an inhibitor of inflammatory and immune responses often associated with chronic autoimmune-based conditions. In New Zealand, the reference adalimumab (Humira) has been listed on the Pharmaceutical Schedule since 2009 and has been funded under Special Authority criteria for use in:4,5
- rheumatoid arthritis
- polyarticular juvenile idiopathic arthritis
- psoriatic arthritis
- ankylosing spondylitis
- Crohn disease
- plaque psoriasis
- hidradenitis suppurativa
- uveitis (ocular inflammation).
Humira is currently used by approximately 6400 patients in the community and is routinely self-administered as a subcutaneous injection. Most patients using Humira who are seen in primary care either have rheumatoid or inflammatory bowel conditions.6
Evidence base and international experience with adalimumab biosimilars
Amgevita, an adalimumab biosimilar, has been approved by the European Medicines Agency (EMA) for use in the EU, by the Food and Drug Administration in the US, by the Therapeutic Goods Administration in Australia, and by Medsafe in New Zealand for all the indications of the reference adalimumab product (Humira).
A European study compared 29 biosimilar monoclonal antibody products and fusion proteins and their reference products (including adalimumab) and demonstrated comparable efficacy, safety and immunogenicity.7 The study used post-marketing surveillance data from European Public Assessment Reports (EPARs) and Periodic Safety Update Reports (PSURs) submitted to the EMA. The study claims that the safety and immunogenicity profiles of the biosimilars and their reference products are similar and do not change upon switching.7
More than 15 years’ use of biosimilars in the EU, over 2 billion treatment days worldwide, and reviews of more than 175 reference/biosimilar switch studies conducted up to 2018 have not revealed any safety problems. “An analysis of more than 1 million patient-treatment years of safety data raised no safety concerns.”7
Two phase III studies, in patients with rheumatoid arthritis8 and in patients with plaque psoriasis,9 both demonstrated non-inferiority between Amgevita and the reference adalimumab.
In Australia, on 1 April 2021, four adalimumab biosimilars were added to the Pharmaceutical Benefits Scheme. Amgevita was one of these and, of the four, it is the biosimilar providing the greatest range of dosage and delivery options (including prefilled syringe and pen, and a paediatric dose); these options are the ones also being made available in New Zealand.10
The 2018 Australian Public Assessment Report (AusPAR) for adalimumab noted that a single-dose pharmacokinetic study demonstrated bioequivalence of Amgevita and Humira.11 AusPAR also concluded that the two aforementioned phase III clinical equivalence studies were “well designed, used appropriate clinical endpoints, adequately justified the equivalence margins and convincingly demonstrated clinical equivalence for patients with rheumatoid arthritis and with plaque psoriasis”.11
New Zealand’s Pharmacology and Therapeutics Advisory Committee (PTAC) concluded that, based on available evidence, biosimilar adalimumab appears equally effective to reference adalimumab with no evidence to indicate any specific clinical risk or harm with switching from the reference product to a biosimilar.12
The committee also considered that there is currently no evidence that the rate of development of immunogenicity – including the identification of treatment antibodies leading to loss of treatment effectiveness – differed between reference and biosimilar adalimumab.12
See the He Ako Hiringa biological medicines resource hub for further reading.
New Zealand's decision to fund Amgevita
In November 2021, following consultation with PTAC12 and a request for feedback on its proposal,4 Pharmac announced a decision to widen access to adalimumab, which will be achieved by funding the biosimilar Amgevita.3 From 1 March 2022, Amgevita (supplied by Amgen) will be funded in New Zealand with a Principal Supply period from 1 October 2022 to 31 July 2026.3
The Special Authority renewal period for Amgevita is being extended to two years and it will be possible for renewals to be requested by any relevant practitioner.2 Compared with Humira, the access criteria for Amgevita have been expanded for existing and new uses, and dosage restrictions have been removed.3 Primary healthcare providers will be able to alert adalimumab-naive patients to the expanded access criteria and, where appropriate, refer for secondary care consultation.
Amgevita will be available in the same dose and delivery options in which Humira has been available (see Table 1):
Expanded access to adalimumab
Adalimumab is used to treat a range of dermatological, rheumatological, gastrointestinal and ophthalmologic conditions. The funding decision and changed criteria brings widened access to adalimumab; it is anticipated that 700 New Zealanders will have improved access to funded adalimumab in the first year.13
On 1 March 2022, the Ministry of Health will automatically issue all current adalimumab patients with an initial Special Authority number for Amgevita. From this date, Amgevita will be funded for all existing uses of adalimumab and for expanded existing and new uses. Dosage limitations will also be removed for Amgevita. (See Table 2)
A seven-month transition window is in place from 1 March 2022 to 30 September 2022, during which time:3
- Amgevita or Humira will both be funded for existing patients and uses*
- patients using Humira should be changed to Amgevita – with some exceptions (see below)
- only Amgevita will be funded for new patients and uses (widened access).
*Ensure any prescription clearly specifies the brand of adalimumab (Amgevita or Humira) to be dispensed.
From 1 October 2022:3
- Amgevita will be the main funded brand of adalimumab for all uses (current and new)
- Humira will remain funded (through a new initial Special Authority – see below) for patients previously treated with Humira who, following discussion with their prescriber:
- have Crohn disease or ocular inflammation and are considered at risk of disease destabilisation if there were to be any change to their treatment regimen.
- trial Amgevita and experience clinical difficulties (intolerable side effects or loss of disease control) and choose to return to Humira.
- All existing Special Authorities for Humira will expire automatically after 30 September 2022. From 1 October 2022, the appropriate prescriber will need to complete a new initial Special Authority for the above patients who require ongoing funded access to Humira.
Table 2: Key information regarding expanded access to adalimumab via prescribing of Amgevita.13
New funded indications from 1 March 2022:
- ulcerative colitis first-line
- undifferentiated spondyloarthritis
- inflammatory bowel disease-associated arthritis.
The following currently funded indications will also have access widened from 1 March 2022:
- Crohn disease dose escalation
- rheumatoid arthritis: reduction in the number of swollen joints required for access to treatment, and removal of the requirement for C-reactive protein level to be >15mg/L
- Behçet disease: access to funded treatment with adalimumab as a first-line biologic
- ocular inflammation: access to funded treatment with adalimumab as a first-line biologic.
Funded access to adalimumab for all indications will be improved by the new Special Authority criteria for Amgevita. These will include:
- removal of dosage restrictions
- extension of Special Authority renewal periods to two years
- allowing any relevant practitioner to apply for Special Authority renewals
- removal of Special Authority renewals for some conditions.
The full Amgevita Special Authority criteria are available on the Pharmac website.14
Transitioning patients from Humira to Amgevita in 2022
Patients using Humira should only receive a first dispensing of Amgevita after having a supporting appointment or discussion with an appropriate prescriber. Primary healthcare professionals involved in the care of patients using Humira should initiate a conversation about transitioning to treatment with Amgevita at the earliest opportunity.
Patients for whom Amgevita might be available under the new or expanded funding criteria should also be informed of this and referred to their specialist, as appropriate, for a discussion about the possible initiation of treatment.
Transitioning stable patients from Humira to Amgevita will benefit from a multi-stakeholder approach by specialists, GPs, primary care nurses and pharmacists; two-way communication between the domains; and knowledgeable and positive conversations with the patient about biosimilars and Amgevita.
As always, the role of each healthcare professional is to ensure that patients receive appropriate treatment and support. The change to adalimumab funding provides additional flexibility in primary care management of patients while maintaining engagement with secondary care as needed.
Everyone in the health system has a role to play to deliver this change, and some specific roles may include:
Specialists: New patients (adalimumab-naive) and patients accessing Amgevita under new or expanded existing indications will continue to have treatment initiated by a specialist. During the transition of stable patients from Humira to Amgevita, specialist support may be sought for:
- patients with additional needs and/or unstable disease
- patients whose disease control has become reduced during the transition
- patients for whom the primary healthcare professional does not feel confident in managing the transition.
General practitioners, nurse practitioners, nurse prescribers, pharmacist prescribers: Stable patients transitioning from Humira to Amgevita may be managed in primary care, which can take responsibility for:
- Amgevita Special Authority renewals (by any relevant practitioner) – renewals will only be required every two years, reducing administrative burden
- the ongoing prescribing of adalimumab in the community
- the referral of patients to relevant specialists where appropriate (eg, additional needs, evidence of disease deterioration)
- helping patients to feel confident and comfortable using Amgevita
- potentially, some training of patients about how to use the Amgevita device
- monitoring for treatment efficacy and adverse effects.
Primary care nurses: Nurses in primary care will have an important role in:
- training patients in the use of the Amgevita device (previously a role performed by secondary care nurses)
- ensuring patients feel confident and comfortable using Amgevita
- monitoring for treatment efficacy and adverse effects.
Community pharmacists: Patients presenting prescriptions for Humira should be given information about the funding changes and prompted to make an appointment to discuss the transition to Amgevita with the appropriate prescriber. The pharmacist has the opportunity also to:
- support the training of patients on how to use the Amgevita device
- ensure patients have received appropriate training at first dispensing of Amgevita
- provide information to promote confidence in Amgevita at first dispensing.
Community pharmacists can initiate the conversation as Humira prescriptions are filled, recommending the patient talk with their prescriber. A brand switch fee will be applied to reimburse pharmacists for their time supporting this change, including time spent providing information to patients.
Using brand names
When prescribing or dispensing biological medicines, the New Zealand Formulary recommends:15
- prescribe by brand name, rather than generic, to avoid inadvertent substitution
- dispense the brand prescribed and record the batch number where possible to ensure the batch is traceable.
In all professional and patient interactions and prescribing, it is essential to refer to the biological medicine by brand name.16
The Special Authorities for Humira and Amgevita are not interchangeable. The access criteria for Humira and Amgevita differ too, and after 30 September 2022 the funding criteria for Humira will change again (and from 1 October 2022, any existing Special Authority numbers for Humira will not be valid). Patients potentially eligible to continue using Humira from 1 October 2022 will need a new initial Special Authority application completed by the appropriate prescriber.
Exemptions that allow use of Humira
In general, based on overseas experience, the transition to Amgevita in primary care following a consultation with an appropriate prescriber is not considered to present difficulties, provided information and support about the change is given.
However, two exemptions allow for certain patients to access Humira:
- Patients with Crohn disease or ocular inflammation who are considered at risk of disease destabilisation with a change in treatment can continue to access Humira.
- Patients on Humira who trial at least two doses of Amgevita (for less than six months after starting Amgevita) and experience clinical difficulty after this trial (intolerable side effects, or loss of disease control attributed to the change) can return to funded treatment with Humira.13
Reporting adverse events
If a patient has a suspected adverse drug reaction to a biological medicine, a report should be submitted to the Centre for Adverse Reactions Monitoring (CARM) at the NZ Pharmacovigilance Centre. All reports should include the brand name and batch number of the suspected biological medicine and can be made via the website: nzphvc.otago.ac.nz/reporting, by email: carmnz@otago.ac.nz or by using a pre-printed card. Electronic reporting is also possible using an adverse reaction reporting tool present in many practice management systems.1
Resources for GPs, nurses and pharmacists
To support the introduction of Amgevita, resource packs for prescribers, nurses and pharmacists will be made available by the supplier, Amgen, and will include:
- clinical information about Amgevita, including efficacy, safety and immunogenicity data
- practical information about Amgevita, including use of the device and effective patient training
- reusable demonstration devices and links to online learning modules
- access to a free medical information phone line to support healthcare professionals with logistical, practical and clinical queries.
Resources for patients
Patients who are prescribed Amgevita will have the following information and support from Amgen:
- key information about Amgevita, device instructions, a patient alert card and information about how to access support services, in a range of languages
- free access to registered nurses (based in Australia) via phone and video to assist with self-administration and medicine queries
- free replacement sharps bins posted directly to patients upon request
- a website with access to supporting documents and videos, and links for ordering sharps bins and contacting remote nursing support.
See the He Ako Hiringa biological medicines resource hub for links to resources.
Patient education, positive conversations and device details
The successful transition of patients from Humira to Amgevita requires interdisciplinary cooperation and consistent messaging by specialists, primary care prescribers, nurses and pharmacists. As an exercise involving biosimilars, it is one that will become increasingly relevant for healthcare professionals as more products come onto the market.
A smooth transition for the patient is dependent on healthcare professionals:
- being a trusted source of knowledge on biologics and biosimilars
- explaining the reasons for, and benefits of, the change
- managing patient anxiety about change
- having positive conversations with patients about Amgevita and promoting expectations of continued disease control
- providing device training.
Good conversations with patients before initiating or changing a medicine help to reduce the likelihood of unwanted outcomes due to the nocebo effect.17 The nocebo effect is a decrease in subjective benefit, a worsening of symptoms or onset of adverse effects due to an expectation or perception of harm associated with a treatment.17
Patient reporting of adverse drug effects is very much related to their expectations and may be affected by how the healthcare professional talks about the medicine. In general, people are much more inclined to pick up on negative rather than positive information, both from outside and online sources, and as hesitancy or doubt conveyed within the healthcare setting.18
Prescribers are instrumental in framing medicines positively or negatively for the patient – prescriber's own beliefs significantly influence how a patient feels about a medicine. A prescriber who is hesitant, diffident or uncertain can transfer these feelings during the consultation, affecting how the patient experiences and accepts a medicine.18,19
A useful approach is to create an environment where the patient can voice their beliefs, and for the prescriber to provide information – perhaps using a health literacy framework to aid the conversation:20
- Ask the patient how they feel about changing from Humira to Amgevita.
- Build on their existing knowledge of the medicines.
- Check with the patient that you have explained things adequately and they know the next steps to be taken.
He Ako Hiringa has produced a bulletin and podcasts about positive framing when initiating new medicines.
Amgevita product characteristics
Amgevita is supplied as a citrate-free formulation21 in a 20mg (paediatric) dose pre-filled glass syringe and as a 40mg dose, in either a pre-filled glass syringe or pre-filled pen.22
Prior to subcutaneous administration, the Amgevita device should be allowed to sit at room temperature for 15–30 minutes. It should not be warmed in any other way. The solution should be inspected closely and not used if it is discoloured, cloudy, or if flakes or particles are present. Vigorous shaking of the product is to be avoided.22
Amgevita does not contain preservatives. Any unused medicine or waste material should be disposed of appropriately.22
The Amgevita pen is highly similar to the Humira device but differs in shape and colour. Both use a clear window that fills yellow over 10 seconds as the injection is delivered. Amgevita product information with a picture of the SureClick pre-filled pen can be found here.21
In using the terms switching, transitioning and substitution, it pays to be clear
Switching is the act by the treating clinician “to exchange one medicine for another with the same therapeutic intent”.16 Switching can refer to a change between two different medicines (eg, infliximab to adalimumab) or between a reference biological medicine and its biosimilar (eg, Humira to Amgevita) or between biosimilars of the same reference product.
Switching from a reference product to a biosimilar (or vice versa) or between biosimilars is also referred to as nonmedical switching (ie, for cost-saving purposes)23 – it has been proposed the term transitioning is used for this type of switching to help delineate the types of switches reported in the literature.23,24
Automatic substitution is where a medicine is dispensed in place of another equivalent medicine that is expected to have the same clinical effect, at the pharmacy level without consultation with the prescriber.16 Note that, in New Zealand, automatic substitution of biological medicines is not permitted.25
Professional development - Assessments for pharmacists, GPs and nurses
Pharmacists - a set of CLASS reflection activities and peer group guides has been prepared in relation to this article, to help you meet your Pharmacy Council annual recertification requirements - download here.
GPs and nurses - you may also find the CLASS activities useful, or you may wish to just complete the self-assessment questions, which you can find in the 'CLASSact answer template' here.
Professional college endorsements
This activity has been endorsed by The Royal New Zealand College of General Practitioners (RNZCGP) and has been approved for up to 1 CME credit for continuing professional development purposes (1 credit per learning hour). To claim your CPD credits, log in to your Te Whanake dashboard and record these activities under the appropriate learning category.
This activity has been endorsed by the PSNZ as suitable for inclusion in a pharmacist’s CE records for CPD purposes.


References
1. bpacnz. Biosimilars: the future of prescribing biological medicines. 31 January 2020. https://bpac.org.nz/2020/biosimilars.aspx
2. He Ako Hiringa. Biosimilars: A promising new era. Bulletin 7, 27 September 2021. www.akohiringa.co.nz/education/biosimilars-a-promising-new-era-bulletin-7
3. Pharmac. Decision to widen access to adalimumab and award Principal Supply. 17 November 2021. https://pharmac.govt.nz/news-and-resources/consultations-and-decisions/2021-11-17-decision-to-widen-access-to-adalimumab-and-award-principal-supply
4. Pharmac. Proposal to widen access to adalimumab and award Principal Supply. 26 August 2021. https://pharmac.govt.nz/news-and-resources/consultations-and-decisions/2021-08-26-proposal-to-widen-access-to-adalimumab-and-award-principal-supply
5. Pharmac. Adalimumab (Humira): Alternative brand access. https://pharmac.govt.nz/assets/2021-11-Alternative-brand-Special-Authority.pdf
6. Pharmac. Request for Proposals – Supply of Adalimumab. 9 March 2021. https://pharmac.govt.nz/assets/rfp-2021-03-09-adalimumab.pdf
7. Kurki P, Barry S, Bourges I, et al. Safety, Immunogenicity and interchangeability of biosimilar monoclonal antibodies and fusion proteins: A regulatory perspective. Drugs 2021;81(16):1881–96. https://doi.org/10.1007/s40265-021-01601-2
8. Cohen S, Genovese MC, Choy E, et al. Efficacy and safety of the biosimilar ABP 501 compared with adalimumab in patients with moderate to severe rheumatoid arthritis: a randomised, double-blind, phase III equivalence study. Ann Rheum Dis 2017 Oct;76(10):1679–87. www.ncbi.nlm.nih.gov/pmc/articles/PMC5629940
9. Papp K, Bachelez H, Costanzo A, et al. Clinical similarity of biosimilar ABP 501 to adalimumab in the treatment of patients with moderate to severe plaque psoriasis: A randomized, double-blind, multicenter, phase III study. J Am Acad Dermatol 2017 Jun;76(6):1093–1102. www.jaad.org/article/S0190-9622(16)31238-5/fulltext
10. NPS MedicineWise. RADAR. Adalimumab biosimilars now PBS-listed. 21 April 2021. www.nps.org.au/radar/articles/adalimumab-biosimilars-now-pbs-listed
11. Australian Government, Department of Health. Therapeutic Goods Administration. Australian Public Assessment Report for Adalimumab. June 2018. www.tga.gov.au/sites/default/files/auspar-adalimumab-180625.pdf
12. Pharmac. Record of the New Zealand Pharmacology and Therapeutics Advisory Committee meeting. 12 and 13 November 2020. https://pharmac.govt.nz/assets/2020-11-PTAC-minutes.pdf
13. Pharmac. Adalimumab (Amgevita): Information for health care professionals. https://pharmac.govt.nz/medicine-funding-and-supply/medicine-notices/adalimumab-healthcare-pros
14. Pharmac. Amgevita access criteria. https://pharmac.govt.nz/assets/2021-11-Amgevita-Special-Authority.pdf
15. New Zealand Formulary. Guidance on medicines use: Biological and biosimilar medicines. https://nzf.org.nz/nzf_70473
16. The European Medicines Agency and the European Commission. Biosimilars in the EU: Information guide for healthcare professionals. © EMA, 2019. www.ema.europa.eu/en/documents/leaflet/biosimilars-eu-information-guide-healthcare-professionals_en.pdf
17. bpacnz. The nocebo effect: what it is, why is it important and how can it be reduced? 30 August 2019. https://bpac.org.nz/2019/nocebo.aspx
18. Petrie K. Legendary Conversations (podcast). Episode One: Initiating new medicines (part 1). He Ako Hiringa. 11 October 2021. www.akohiringa.co.nz/education/episode-one-initiating-new-medicines-part-1
19. He Ako Hiringa. Starting a medicine? Accentuate the positive. Bulletin 9, 26 November 2021. www.akohiringa.co.nz/education/starting-a-medicine-accentuate-the-positive
20. Health Quality & Safety Commission New Zealand. Three steps to better health literacy – a guide for health care professionals. 18 November 2021. www.hqsc.govt.nz/resources/resource-library/three-steps-to-better-health-literacy-a-guide-for-health-care-professionals
21. Amgen (New Zealand) Limited. Launch mailer (PDF): What's behind Amgevita makes the difference. January 2022. https://pharmac.govt.nz/assets/AM10660-Amgevita-Launch-Mailer-1-v3.pdf
22. Medsafe. New Zealand data sheet. Amgevita. Available online at www.medsafe.govt.nz/profs/Datasheet/a/amgevitainj.pdf
23. Barbier L, Ebbers HC, Declerck P, et al. The efficacy, safety, and immunogenicity of switching between reference biopharmaceuticals and biosimilars: A systematic review. Clin Pharm Ther 2020;108(4):734–55. https://ascpt.onlinelibrary.wiley.com/doi/full/10.1002/cpt.1836
24. Dörner T, Kay J. Biosimilars in rheumatology: current perspectives and lessons learnt. Nat Rev Rheumatol 2015;11:713–24.
25. Medsafe. Biosimilars. 2014. Available at: www.medsafe.govt.nz/profs/RIss/Biosimilars.asp Accessed December 2021.